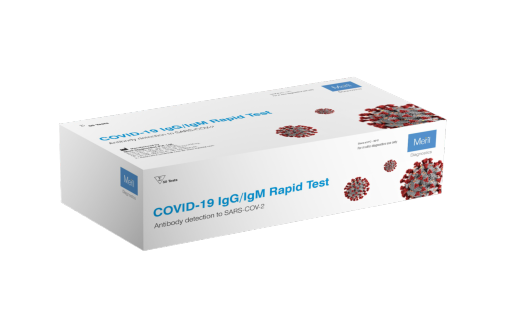
MERIL COVID-19 IGG/IGM RAPID TEST KIT
MERIL COVID-19 IgG/IgM Rapid Test is an Immunochromatographic assay for rapid, qualitative differential detection of antibodies (IgG & IgM) against Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) in human whole blood or serum/plasma sample. The test is to be used as an aid in the diagnosis of coronavirus infection disease (COVID-19) which is caused by SARS-CoV-2. The test provides preliminary test results. Negative results do not preclude SARS-CoV-2 infection and they cannot be used as the sole basis for treatment.
Click here to know more about Covid-19 testing solutions
- Based on Lateral Flow Technology
- Diffrential Detection of IgG and IgM antibodies against SARS COV 2
- Highly purified Antigens are conjugated for higher accuracy
- Sample volume: 10 μL of Whole blood/Serum/Plasma
- Test Result in 20 minutes
- Sensitivity: 97.20% & Specificity: 99.22%
Benefits
Indication
- Identification of current or past infection
- Best suited for quick screening
Product Specifications
| Intended Use | Differential detection of IgG & IgM antibodies against SARSCoV-2 |
| Principle | Immunochromatography |
| Technique | Lateral Flow |
| Detection | IgG and IgM antibodies against SARS COV 2 |
| Coated with | Highly purified Antigens are conjugated for higher accuracy |
| Type of Specimen required | Whole Blood/ Serum/ Plasma |
| Specimen Volume | 10 μL |
| Buffer Volume | 3 drops |
| Result interpretation time | 20 minutes |
| Diagnostic Sensitivity | 97% |
| Diagnostic Specificity | 99.22% |
| Evaluations | Indian Council of Medical Research (ICMR) |
Ordering Information
| Product Code | Material Description | Pack Size |
|---|---|---|
| NCVRPD-01 | MERIL COVID-19 IgG/IgM Rapid Test | 25 Tests |
| NCVRPD-02 | MERIL COVID-19 IgG/IgM Rapid Test | 50 Tests |
| NCVRPD-03 | MERIL COVID-19 IgG/IgM Rapid Test | 10 Tests |
To download IFU in German Language, Click here
Click on the below documents name to view/download the same:
Clinical Data
Product IFU
Note: IFU will be displayed after MDR Certification